|
The following six reactions represent common cathodic reactions along with the conditions in which they generally occur: 1. I can imagine copper hydroxide being precipitated at the anode and $NO$ gas being liberated there as well, but it could get more complicated, as $NO$ reacts with $OH^-$. The reduction half-cell reaction at the cathode depends mainly on environmental conditions. The possible cathode reactions can be classified as follows: Cathode reactions Disproportionation Topochemical Abstraction Insertion I Intercalation A disproportionation reaction here is one such as is found with the copper halides CuF2 + 2Li -> 2LiF -t- Cu where the crystalline structure of the cathode is com. The pH will increase because $H^+$ is consumed.Īt the cathode, copper will plate out, not oxygen. During discharging, the lithium ions travel back from the anode to the cathode, in the process giving up. 
Electrolyzers using a liquid alkaline solution of sodium or. membrane, to the negative anode electrode. Alkaline electrolyzers operate via transport of hydroxide ions (OH-) through the electrolyte from the cathode to the anode with hydrogen being generated on the cathode side. In any electrochemical cell, the electrons always move from anode to cathode. But nitrate ion is a a bit more active than a mere spectator ion and some will be reduced at the anode to $NO$. Anode Reaction: 2H 2 O O 2 + 4H + + 4e-Cathode Reaction: 4H + + 4e- 2H 2 Alkaline Electrolyzers. Galvanic cells always involve spontanous oxidation-reduction reactions. Not all half-reactions must be balanced with acid or base. 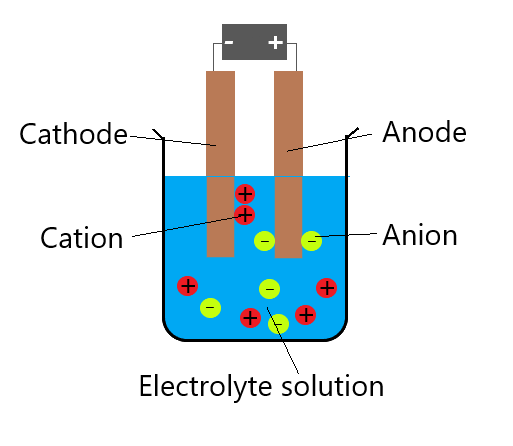
The same half-reactions can also be balanced with the base as listed below. There are two copper blocks sitting in the $\ce$ in the solution is driven onto the cathode to maintain neutrality everywhere. Indeed, one feature that sets apart the DMFC is that there is considerable voltage loss at both the anode and the cathode. At the positively charged anode, an oxidation reaction occurs, generating oxygen gas and giving electrons to the anode to complete the circuit: Oxidation at anode: 2 H 2 O O 2 (g) + 4 H + (aq) + 4e.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
